According to the National Cancer Institute, a biomarker is “a biological molecule found in blood, other body fluids, or tissues that is a sign of a normal or abnormal process, or of a condition or disease,”(NCI) such as cancer. Biomarkers typically differentiate an affected patient from a person without the disease.
Table of Contents
What is biomarker?
“A biomarker — short for “biological marker”—is anything that can be assessed objectively and is either a sign of a normal or abnormal procedure, or a disease or illness. A biomarker may be a molecule present in the blood, or other tissue or body fluids.
Another form of biomarker is a genetic signature or “fingerprint” — an activity pattern in a collection of genes that indicates a certain biological condition.
Examples of biomarkers cover everything from pulses and blood pressure to simple chemistries to more complicated blood and other tissue tests in the laboratory.
In oncology, a biomarker may be a molecule that is secreted by a tumor, or a body ‘s specific reaction to cancer.
This may help to detect cancers in the early stages, forecast how dangerous a cancer can be or predict how well a patient may respond to treatment.
Some examples of biomarkers used for cancer screening or treatment effectiveness tests include AFP (liver cancer), BCR-ABL (chronic myeloid leukemia), CA-125 (ovarian cancer), CA19.9 (pancreatic cancer) and CEA (colorectal cancer).
Biomarkers also help to predict or control recurrence of cancer. For example, the Oncotype DX ® test on breast cancer is used to determine the risk of recurrence of breast cancer in women being treated for early breast cancer.
Oncotype DX tests a set of 21 genes in cells taken during tumor biopsy, and offers a risk score for recurrence.
Additionally, cancer biomarkers have demonstrated usefulness in measuring how well a drug performs over time. Researchers continue to explore the potential of these biomarkers as alternatives to current image based research, such as CT scans and MRIs.
One of biomarker research’s strongest fields is immunotherapy, a form of treatment that manipulates a patient’s immune system to combat drug cancer or modified immune T cells.
In recent years, immunotherapy has had significant success in certain patients with some forms of cancer, but as of now, only a minority of patients are responding to treatment with immunotherapy agents.
Researchers are working hard to find biomarkers that could identify, which patients are likely to respond to immunotherapy in advance. In some forms of cancer, a better or worse response to immunotherapy was associated with the presence or absence of immune “checkpoint” molecules, such as PD1 on immune cells or PDL-1 in cancer cells.
For example, one clinical trial showed that the immunotherapy drug pembrolizumab lowered the risk of death in patients with non-small cell lung cancer – and whose tumors expressed high levels of PDL-1—by 37 percent compared to chemotherapy.
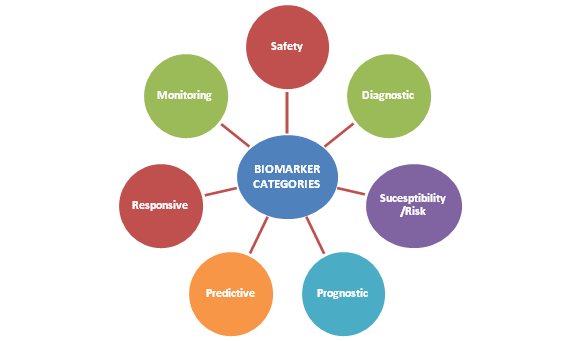
What are the ![]() types of Biomarkers?
types of Biomarkers?
file:///C:/Users/hp/Downloads/33f111bfadb61211e9775dbf48c843c92f4220f9.pdf
Biomarkers can also be classified as:
- Molecular – have biophysical properties, which allow their measurements in biological samples Eg, Blood glucose plasma, serum, cerebrospinal fluid, bronchoalveolar lavage, biopsy
- Radiographic – obtained from imaging studies
Eg: Bone mineral density
- Histologic – reflect biochemical or molecular alteration in cells, tissues or fluids
Eg: Grading and staging of cancers
- Physiologic – measures of body processes
Eg: Blood pressure
Biomarkers can indicate a variety of health or disease characteristics, including the level or type of exposure to an environmental factor, genetic susceptibility, genetic response to environmental exposure, markers of subclinical or clinical disease, or indicators of response to the therapy.
Applications of Biomarkers
Biomarkers have many useful applications in health care, including prevention and diagnosis of diseases, risk assessment for specific diseases, and monitoring of diseases. They may also be used to assess the safety or toxicity of a treatment procedure, or other exposures to the environment.
| Applications of Biomarkers | ||
| Biomarker Application | Biomarker Examples | Disease state |
| Screening | Prostate specific antigen (PSA) | Prostate cancer |
| Fecal occult blood test | Colon cancer | |
| Susceptibility/Risk | Breast cancer genes 1 and 2 mutations (BRCA 1/2) | Predisposition to developing breast cancer |
| Factor V Leiden | Predisposition to developing thromboembolism | |
| Apolipoprotein C | Predisposition to developing Alzheimer’s disease | |
| Human papillomavirus (HPV) | Predisposition to developing cervical cancer | |
| Diagnostic | Troponin-I | Coronary ischemia |
| Sweat chloride | Cystic fibrosis | |
| Ejection fraction (EF) | Cardiomyopathy/congestive heart failure | |
| Glomerular filtration rate (GFR) | Chronic kidney disease | |
| Prognostic | BRCA ½ | Likelihood of second cancer in women with breast cancer |
| Chromosome 17p deletions and TP53 mutations | Likelihood of death in patients with chronic lymphocytic leukemia | |
| Monitoring | Serum low-density lipoprotein (LDL) | Response to lipid lowering drugs |
| International Normalized Ratio (INR) | Efficacy of anticoagulant therapy | |
| Cancer antigen 125 (CA-125) | Ovarian cancer disease status or burden | |
| Hemoglobin A1C | Response to antihyperglycemic agents or lifestyle changes | |
| Viral load | Response to antiretroviral treatment for human immunodeficiency virus (HIV) | |
| Safety | Hepatic aminotransferases and Bilirubin | Hepatotoxicity |
| Serum creatinine | Nephrotoxicity | |
| Serum potassium | Hypo or Hyperkalemia while taking diuretics, angiotensin converting enzyme inhibitors | |
| Corrected QT interval (QTc) | Assess potential for drugs to induce ventricular tachycardia | |
| Serum Bisphenol A (BPA) | Measured in pregnant women; predicts adverse birth outcomes such as preterm birth; has informed/improved public safety through BPA-free plastics. |
The use of validated biomarkers has become widespread both in basic and clinical science and in clinical practice, and their use as endpoints in clinical trials is now generally recognized.
Biomarkers allow for better understanding disease processes and the ways in which drugs work to fight disease. This information may be used earlier to treat, or to avoid, illness before it starts.
Biomarkers may be used to boost the effectiveness and safety of current drugs, and to produce new drugs. New molecular biomarkers have the ability to tailor disease prevention and treatment, making healthcare delivery more reliable, safe and cost-effective and enhancing patient outcomes.”
References:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3078627/
https://www.niehs.nih.gov/health/topics/science/biomarkers/index.cfm
https://www.sciencedirect.com/topics/neuroscience/biomarkers
https://www.sciencedirect.com/topics/medicine-and-dentistry/biological-marker
https://www.fda.gov/drugs/cder-biomarker-qualification-program/what-are-biomarkers-and-why-are-they-important-transcript
https://www.nursingcenter.com/ncblog/november-2019/biomarker
file:///C:/Users/hp/Downloads/BIOMARKER-TERMINOLOGY–SPEAKING-THE-SAME-LANGUAGE.pdf
https://blog.dana-farber.org/insight/2018/10/what-is-a-biomarker/
file:///C:/Users/hp/Downloads/33f111bfadb61211e9775dbf48c843c92f4220f9.pdf


